Your second method is the exact same process, except you deal in enthalpy. This is your first method, and it is the correct answer. 
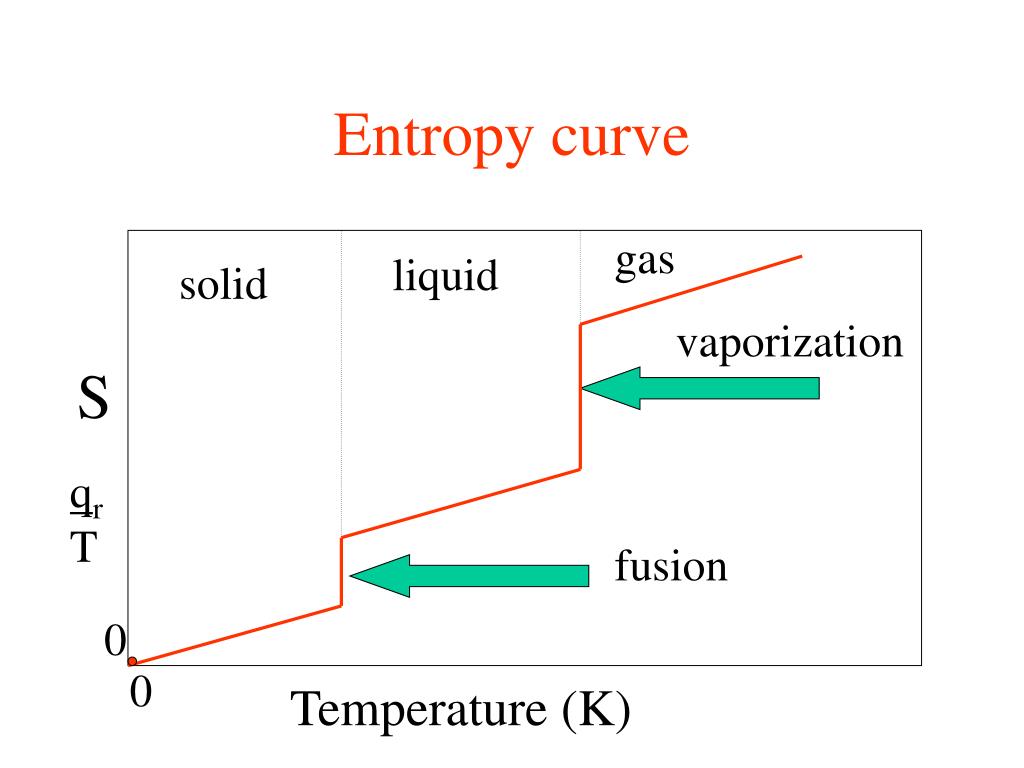
subtract the change in entropy as the vapor is cooled from 100 deg C to 85 deg C. sum the change in entropy from 85 degC to 100 deg Cģ. If someone could tell me what I did wrong, I would really appreciate it! I thought to find the :delta: H vaporization at 100☌ by doing 373.15K *109.0 J/K*mol = 40673.35 J/mol. However, I initially did it a different way and wanted to know what was wrong with what I did: The answer obtained from this method is 111 J/K*mol. I realize that it is possible to do this problem by finding the entropy change to heat liquid water from 85☌ to 100☌, finding the entropy change in vaporizing this liquid water (which would just be 109.0J/K*mol), and finding the entropy change to lower the temperature of the water vapor back to 85☌. Calculate the standard entropy of vaporization of water at 85☌, given that its standard entropy of vaporization at 100.☌ is 109.0 J/K*mol and the molar heat capacities at constant pressure of liquid water and water vapor are 75.3 J/K*mol and 33.6 J/K*mol, respectively, in this range. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
